Question Video: Identifying an Unknown Solution from the Reaction with a Sodium Sulfite Solution | Nagwa
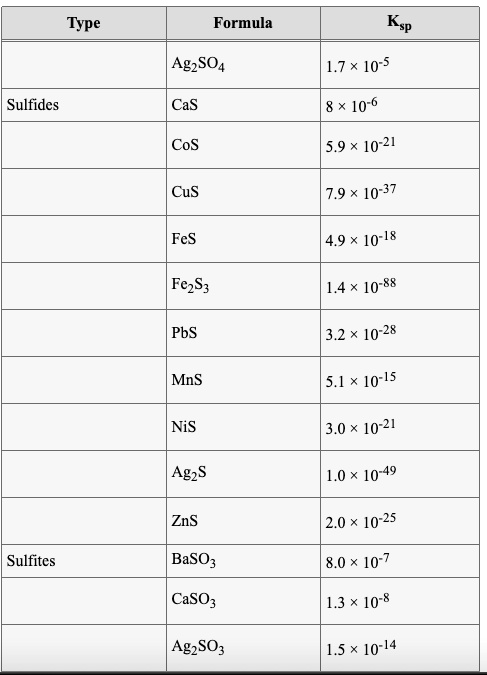
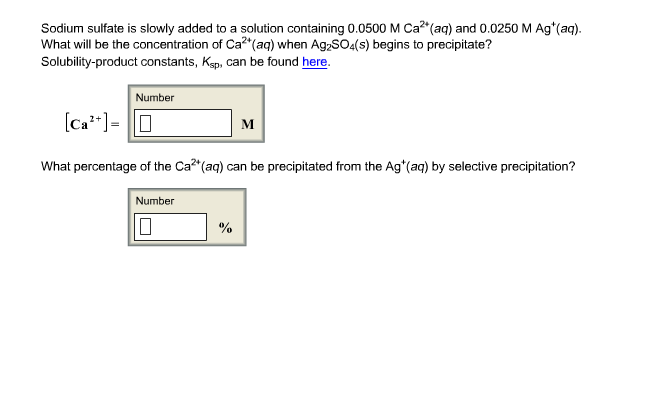
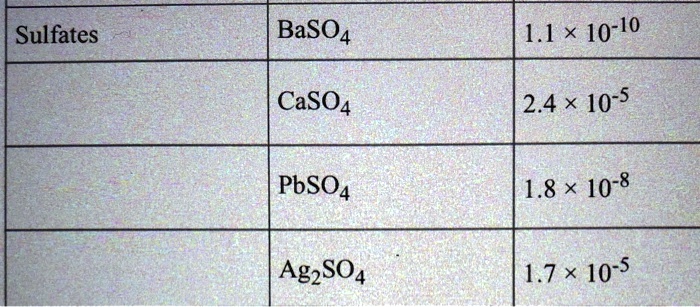
SOLVED: Formula Ag2SO4 L.7 x 10^-5 Sulfides CaS 8 * 10^-6 CoS 5.9 x 10^21 CuS 7.9 x 10^-37 FeS 4.9 x 10^-18 FczS 1.4 x 10^-88 PbS 3.2 x 10^-28 MnS

In the following reaction sequence in aqueous solution, the species X, Y and Z, respectively, are S2032- Ag Ag with time 9 X - Y Clear white black solution precipitate precipitate (A) (
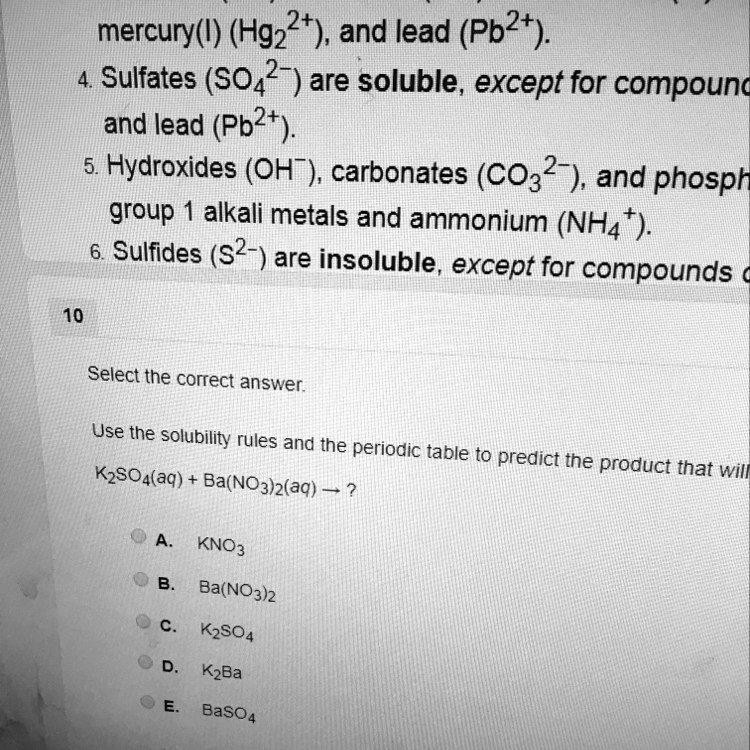
SOLVED: Use the solubility rules and periodic table to predict the product that will precipitate out in the reaction of mercury(I) (Hg2^2+), and lead (Pb^2+) sulfates (SO4^2- are soluble, except for compounds
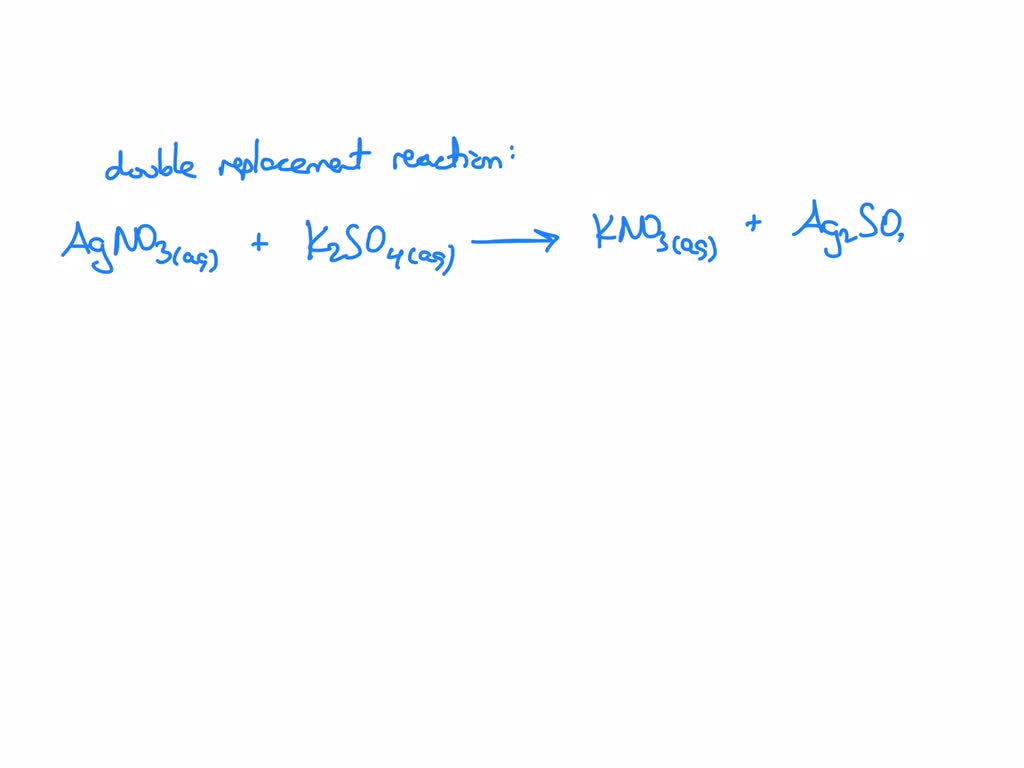
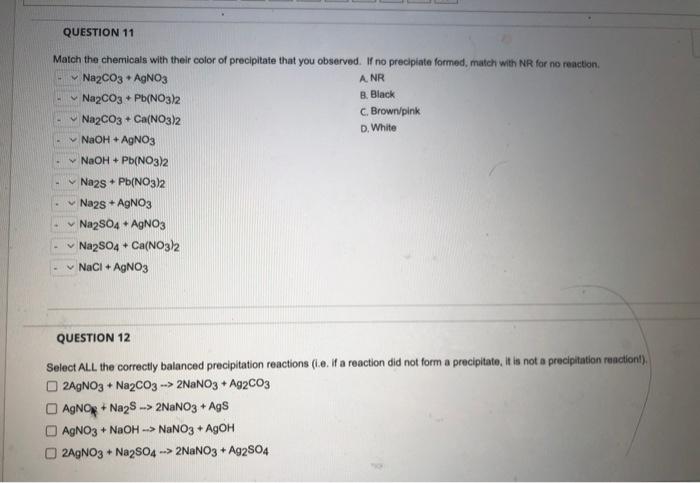
SOLVED: What occurs when aqueous silver nitrate, AgNO3, reacts with aqueous potassium sulfate, K2SO4?' Select one: Ag2SO4 forms as a precipitate. KNO3 forms as a precipitate. K2SO4 forms as a precipitate. No

Why does the color of Silver sulphate NPs change from yellow to dark violet without change to red color? | ResearchGate

Transformation from Ag@Ag3PO4 to Ag@Ag2SO4 hybrid at room temperature: preparation and its visible light photocatalytic activity | SpringerLink

SOLVED: Precipitation of Ag2SO4 Chemical equation: 2 Ag(aq, 0.025 M) + SO4 (aq, 0.025 M) â†' 3 Ag2SO4(s) ΔG° = -28.094 kJ/mol ΔG°mn,298 K = ΔG°TIn,298 K + 2.303 RT log Q