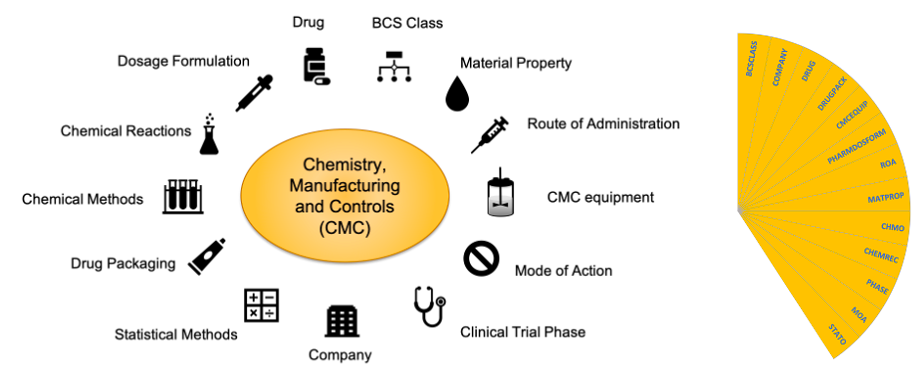
Transitioning Chemistry, Manufacturing, and Controls Content With a Structured Data Management Solution: Streamlining Regulatory Submissions - Journal of Pharmaceutical Sciences
APPENDIX 8 CHEMISTRY, MANUFACTURING AND CONTROLS REQUIREMENTS FOR CELL, TISSUE OR GENE THERAPY PRODUCTS FOR CLINICAL TRIALS A

Chemistry, Manufacturing, and Controls (CMC) and Good Manufacturing Practices (GMPs): The Big Picture of a Long-term Commitment Elizabeth Pollina Cormier, - ppt video online download

Innovation in Chemistry, Manufacturing, and Controls—A Regulatory Perspective From Industry - ScienceDirect

Transitioning Chemistry, Manufacturing, and Controls Content With a Structured Data Management Solution: Streamlining Regulatory Submissions - ScienceDirect

Emerging Strategies for Drug-Product Comparability and Process Validation: Part 2 - BioProcess InternationalBioProcess International
![PDF] Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar PDF] Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d2444156a193593569940cb4cfe117fa25349da7/5-Figure3-1.png)
PDF] Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar

Quality Control Made Simple: Understanding Chemistry and Manufacturing Controls | by Max Wilson | Medium

Professional Pharma Solutions LLC " PPS" - Chemistry, Manufacturing and Controls (CMC) ensures that pharmaceutical and biopharmaceutical drug products are consistently effective, safe and high quality for consumers. Regulatory requirements and expectations
![PDF] Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar PDF] Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d2444156a193593569940cb4cfe117fa25349da7/4-Figure2-1.png)
PDF] Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar

Continued Process Verification: Evolution of Biopharmaceutical Control Strategy - CMC ForumBioProcess International

![cmc [ chemistry manufacturing control ] | PPT cmc [ chemistry manufacturing control ] | PPT](https://image.slidesharecdn.com/presentation2222ra-181120122336/85/cmc-chemistry-manufacturing-control-1-320.jpg?cb=1665731435)