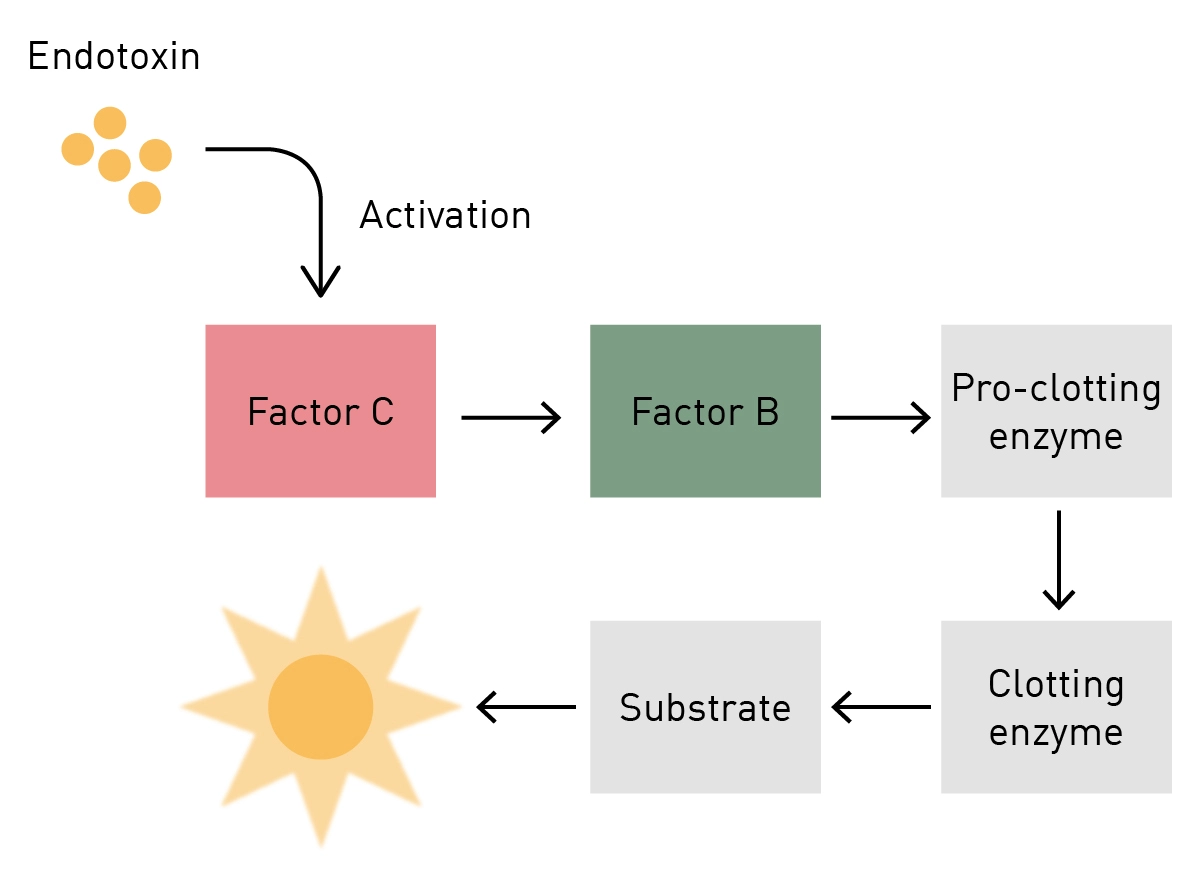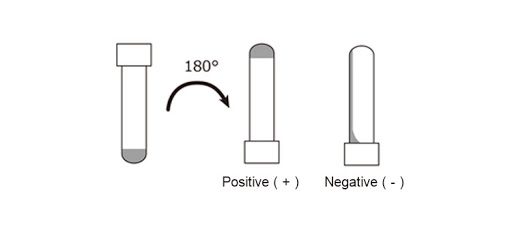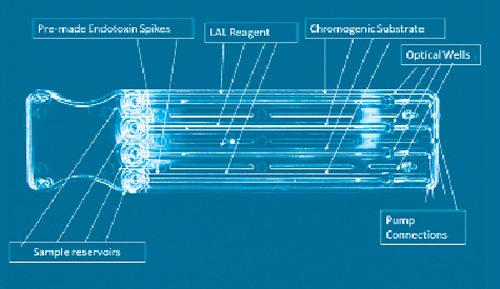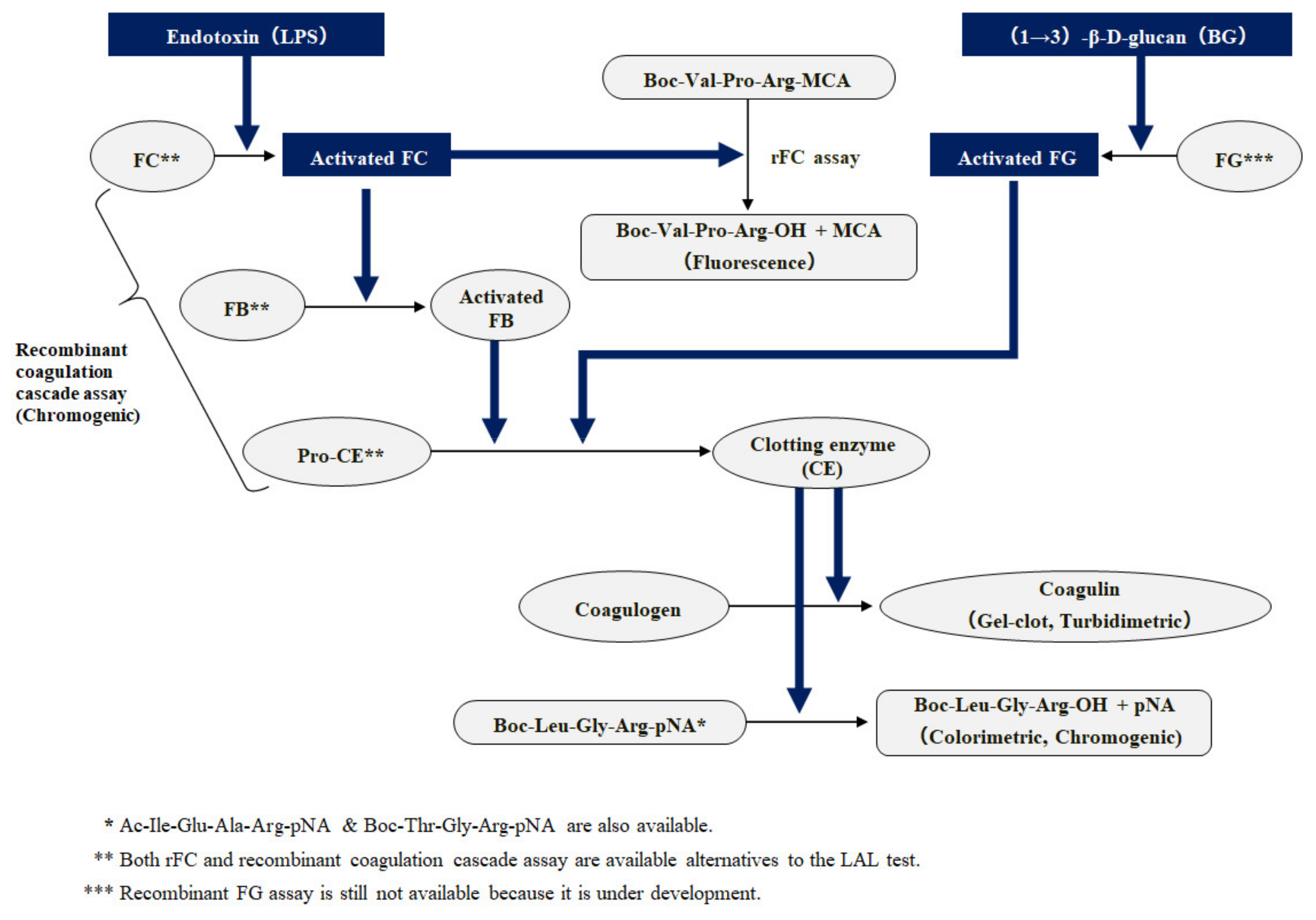
Biomedicines | Free Full-Text | Outstanding Contributions of LAL Technology to Pharmaceutical and Medical Science: Review of Methods, Progress, Challenges, and Future Perspectives in Early Detection and Management of Bacterial Infections and

LAL test validation protocol flow-chart. The test was performed three... | Download Scientific Diagram

Test LAL, test dell'endotossina, reagente LAL, test del coagulo di gel, test del fattore C ricombinante, test dell'endotossina batterica, rilevamento dell'endotossina, produttori e fornitori di indicatori di endotossina - Tecnologia Bioendo